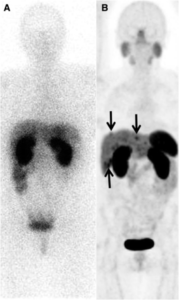
Ga-68 Dotatate PET/CT scan
If you’ve ever been tested for eyeglasses, you know how one lens can create grainy, foggy images and the next crisp, clear letters. That’s the kind of difference you can see between the Ga-68 PET/CT scan and prior technology. But are these newer imaging techniques better? And who stands to benefit most from them?
Ga-68 dotatate is a radiopharmaceutical tracer used during PET (positron emission tomography) scans. Some PET/CT combination scanners also run a CT (computed tomography) scan in the same session and then merge the images together. The Ga-68 PET/CT full-body scan can capture neuroendocrine tumors (NETs) that overexpress somatostatin receptors and show where the tumors are in the body.
What is a Ga-68 PET/CT scan?
“The Ga-68 PET/CT scan is a tool for the care of low or intermediate grade neuroendocrine tumors, primarily for making treatment decisions with early detection of small lesions that impact treatment plans,” said Renuka Iyer, MD, Co-Director of the Liver and Pancreas Tumor Center at Roswell Park Comprehensive Cancer Center, Buffalo, NY.

Dr. Iyer considers the Ga-68 dotatate PET/CT scan to be superior to an octreotide scan—that uses Indium-pentetreotide—which has been the standard of care. “The octreotide scan is less sensitive, and for many more disease is detected with Ga-68 PET/CT.”
The results inform treatment planning. “Having a Ga-68 PET/CT scan that shows no signs of disease beyond a primary tumor and resectable metastatic cancer would support an aggressive surgery approach as opposed to using systemic therapies if more disease is seen, according to Dr. Iyer. She also said Ga-68 PET/CT scan results may help predict response to treatments that rely on somatostatin receptors.
This newer scan, however, is not always the best scan in every situation. “Ga-68 can replace octreotide-based full-body scans, but it does not replace the essential role of conventional CT and MRI scanning for monitoring disease and for those who are not receptor positive,” said Josh Mailman, NETRF treasurer, and a contributing author to the paper “Appropriate Use Criteria for Somatostatin-receptor Imaging” published in the Journal of Nuclear Medicine.
How does a Ga-68 PET/CT scan work?
Published reports show about 70-90 percent of neuroendocrine tumors have somatostatin receptors. Think of this receptor as a docking station for the hormone somatostatin. It’s a place for it to land and connect to the cell. In healthy cells, these receptors are part of signaling system to regulate hormone secretions. For NETs that overexpress hormones, somatostatin receptors serve as a target for therapy given by oncologists and nuclear radiologists. Synthetic somatostatin analogs, paired with radioactive agents, can bind to somatostatin receptors. This radiopharmaceutical can light up neuroendocrine cancer cells in a nuclear scan.
The more uptake in a Ga-68 PET/CT scan, the better the outcome may be with other therapies that target somatostatin receptors. “PRRT is well correlated with activity and uptake of Ga-68 in scans,” said Dr. Iyer.
What is it like to have a Ga-68 PET/CT scan?

Paul underwent a Gal-68 scan at Roswell Park. He asked his doctor for the scan because he had adverse reactions to traditional CT scans. For three to four days after a CT scan, he would experience chills, shakes, stiffness, and soreness. He also didn’t like the taste of the contrast material he had to drink for CT scans.
“I was worried they would see more cancer, but that wasn’t the case,” he said.
Paul’s results showed good uptake of Ga-68 and helped confirm he was a candidate for PRRT. He underwent PRRT over the summer. “I’ve been great, feeling better than last year,” said Paul. At 63 he is now riding his bike and swimming regularly.
Where can you get a Ga-68 scan?
The Ga-68 scan is not offered at every cancer center. Northern California (NorCal) CarciNET Community, a NET patient support and advocacy group, offers a searchable database to help patients find centers.
Roswell Park began offering the Ga-68 PET scan in 2017. According to Dr. Iyer, Roswell Park has not had any access issues. “We don’t have a waiting list or any delays for Ga-68 PET scans or PRRT,” she says.
Is the Ga-68 scan covered by insurance?
Though Paul said he didn’t have trouble getting insurance coverage for his Ga-68 scan, other patients have faced challenges. Aetna announced coverage in August of 2018, following a recommendation by the Society of Nuclear Medicine and Molecular Imaging. NorCal CarciNet offers tips to help patients and families obtain insurance coverage.
Learn more about NET imaging
- Hear Sumeet Virmani, MD, Rush University, give a good overview of Ga-68 https://youtu.be/W-BMSkOpQTU
- Hear Richard Wahl, MD, Washington University (St. Louis), presents an overview of the most promising nuclear medicine diagnostic and therapeutic approaches including Ga-68. https://youtu.be/AIjAE1p-5F8
- Hear Andre Iagaru, MD, Stanford University, talk about his institution’s experience with Ga-68 since 2013. https://youtu.be/FOUg3t7llms
- Not all NETs have somatostatin receptors. Learn about an innovative approach using amino acids to image NETs under investigation by NETRF-funded researcher Babak Behnam Azad, PhD, Johns Hopkins https://netrf.org/a-novel-agent-for-imaging-nets/
- Read about appropriate use criteria for somatostatin-receptor imaging in NETs. http://www.snmmi.org/ClinicalPractice/content.aspx?ItemNumber=23260
Everything you need to know about NET imaging
Listen to this NETWise podcast episode to understand the difference between MRI, CT, and Ga-68 dotatate scans. You’ll understand what to expect from each of these scans and be better able to talk to your doctor about what is best for you.
Share this page on social media