Lung neuroendocrine tumors (NETs) are graded as typical or atypical carcinoids based on how quickly the tumor cells divide. They exhibit diverse clinical behaviors and represent 25% of all NETs. Molecular analyses have identified different groups, ranging from less aggressive to more aggressive forms, including a group termed ‘supra-carcinoids’ that combine low-grade histopathological features with a high-grade molecular profile, leading to poorer survival outcomes.
More research is needed to understand and predict which groups of lung NETs may become more clinically aggressive so that treatment plans can be personalized. However, a shortage of preclinical models has hindered basic NET research, which limits the understanding and development of potential therapeutic targets and mechanisms.
Dr. Hans Clevers, MD, PhD of the Hubrecht Institute in the Netherlands was awarded a 2017 NETRF Petersen Accelerator Award to develop tumor models such as patient-derived tumor organoids (PDTOs) to model NETs and screen for therapeutic vulnerabilities. PDTOs are 3D cultures of patient-derived tumor cells designed to mimic tumor behavior so that they can be studied in the lab.
Dr. Clevers and his team recently published their success in creating tumor organoids in the journal Cancer Cell. They described how they established a diverse collection of lung NET PDTOs, providing a robust platform for research. They further found that the PDTOs maintain the characteristics of the original tumor, such as similar histological and molecular features. This suggests that these models can be used to replicate lung NETs’ biology, to learn more about how they grow, and to test therapeutics.
The lung PDTOs revealed that a subset of lung NETs depend on the growth factor epidermal growth factor, EGF, pointing toward new treatment avenues. Their findings suggest that targeting growth-factor-mediated pathways could be a promising strategy for treating specific types of NETs. Identifying EGF dependence in some lung NET PDTOs suggests the potential use of treatments targeting the epidermal growth factor receptor, EGFR, or its downstream signaling pathways, which are already in use for other tumor types.
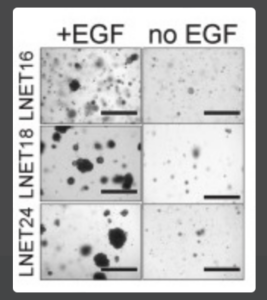
Figure showing the growth of lung NET PDTOs in the presence or absence of EGF, 31 days (LNET16), 42 days (LNET18), or 75 days (LNET24) after plating. From Dayton et al., 2023, Cancer Cell 41, 2083–2099. https://doi.org/10.1016/j.ccell.2023.11.007. Published under the terms of the Creative Commons Attribution – NonCommercial – NoDerivs (CC BY-NC-ND 4.0).
“In generating these tumor organoid models, we discovered that some lung NETs are dependent on the growth factor, EGF, to grow. If we inhibit the receptor for EGF, EGFR, the EGF-dependent organoids die,” says Dr. Talya Dayton, co-first author of the paper published in Cancer Cell. “While further research is needed to confirm these findings, this may mean that patients with lung NETs that express EGFR could be treated with inhibitors of EGFR. Because EGFR inhibitors are already commonly used to treat other kinds of tumors, this could have a significant impact on patients with lung NETs.”
This study demonstrates the immense potential of PDTOs in understanding and treating NETs. By identifying vulnerabilities and testing responses to various treatments, we can move closer to personalized and more effective therapies for NETs. The Neuroendocrine Tumor Research Foundation is proud to have funded this research and remains committed to advancing research and improving outcomes for patients affected by neuroendocrine cancers.
