Home » For Patients » Treatment of NETs » Nuclear Medicine Therapy
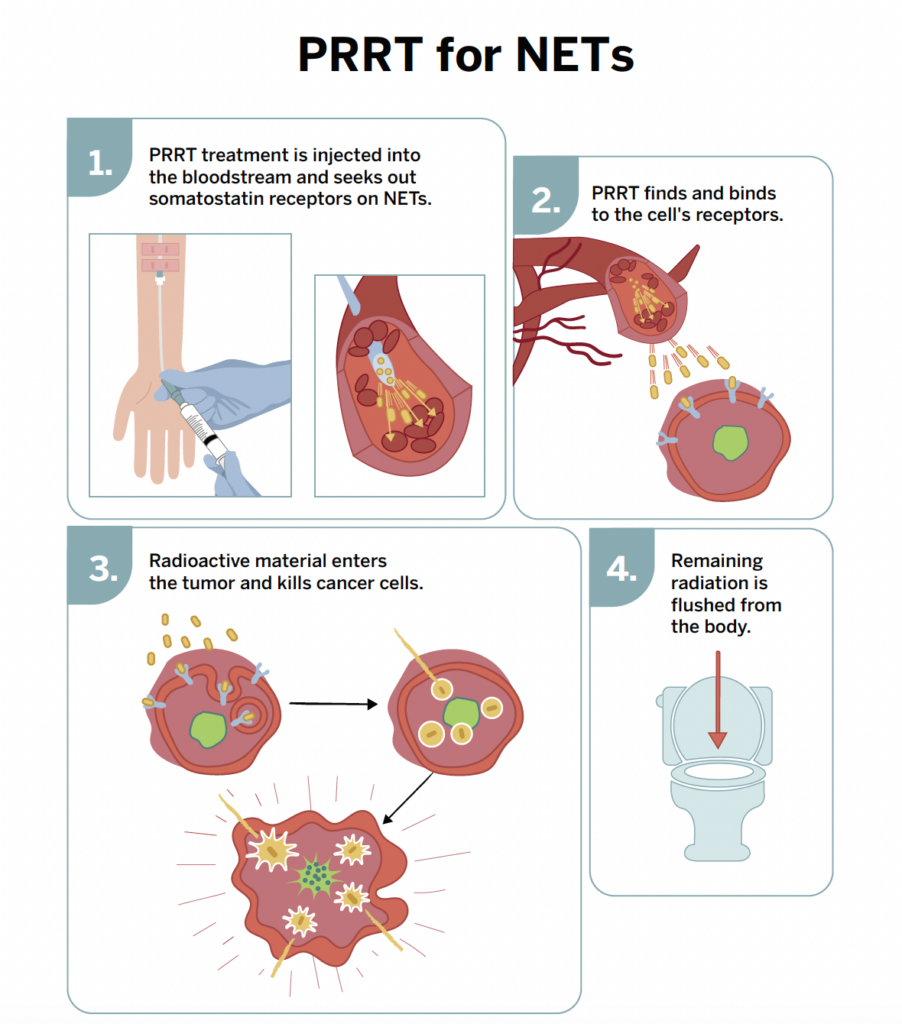
Radioactive drugs to treat neuroendocrine tumors have been available in Europe for many years, and peptide receptor radionuclide therapy (PRRT) with lutetium Lu 177 dotatate was approved in the European Union in 2017. In 2018, the U.S. FDA approved lutetium Lu 177 dotatate for adults with somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs), and later that year approved iobenguane I 131 for patients with unresectable, locally advanced, or metastatic pheochromocytoma or paraganglioma that requires systemic anticancer therapy. These drugs are given intravenously, but they target tumors in different ways. PRRT, also called a form of radioligand therapy, uses radiolabeled peptides to bind somatostatin receptors on tumor cells and deliver radiation directly to the cancer.
PRRT is a treatment that delivers radiation directly to tumor cells through the bloodstream using radiolabeled peptides. Many neuroendocrine tumor cells express high levels of somatostatin receptors on their surface. PRRT uses synthetic peptides that bind to these receptors and carry a radioactive isotope. Once the peptide binds to the tumor cell, it is taken up into the cell, where the attached radioisotope releases radiation that damages and kills the cancer cells while limiting exposure to surrounding healthy tissue.
Studies of Lu-177 have shown PRRT can:
Although generally well tolerated, PRRT with Lu-177 may have side effects such as:
These common side effects are often mitigated or avoided by taking amino acids, fluids, and antinausea medication at the time of treatment. Less common and more serious side effects include bone marrow or kidney toxicity. Studies have shown there is a risk of less than 2% of developing blood cancers such as leukemia or myelodysplastic syndrome.
NETRF has funded research to study new nuclear particles to be used in PRRT, as well as combinations of PRRT and existing drugs that may make the treatment more effective.